Both of these concepts, however, rely on the principles of nuclear charge and the ability to attract electrons.Įvery element has a different electron affinity. Electronegativity is the measure of how much an atom will attract a pair of electrons in a bond.
Electron affinity is related to electronegativity, but is a quantitative measure of the energy change (in kilojoules per mole) of an atom when an electron is added. Electron affinity is the opposite of ionization energy (ionization energy is the energy required to remove an electron from an atom). This property is measured when the elements are in a gaseous state only - this is because it is measured with the atoms sufficiently separated so as to not have their energy levels be affected by surrounding atoms. Both of these concepts, however, rely on the principles of nuclear charge and the ability to attract electrons.Įlectron affinity is the measure of the energy released when an electron is added to an atom to create a negative ion. Is electron affinity the same as electronegativity? Of the metals, mercury has the lowest electron affinity.ģ.
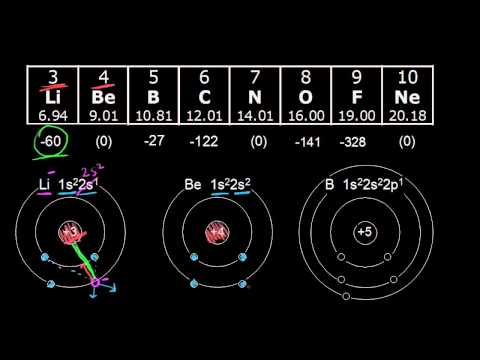
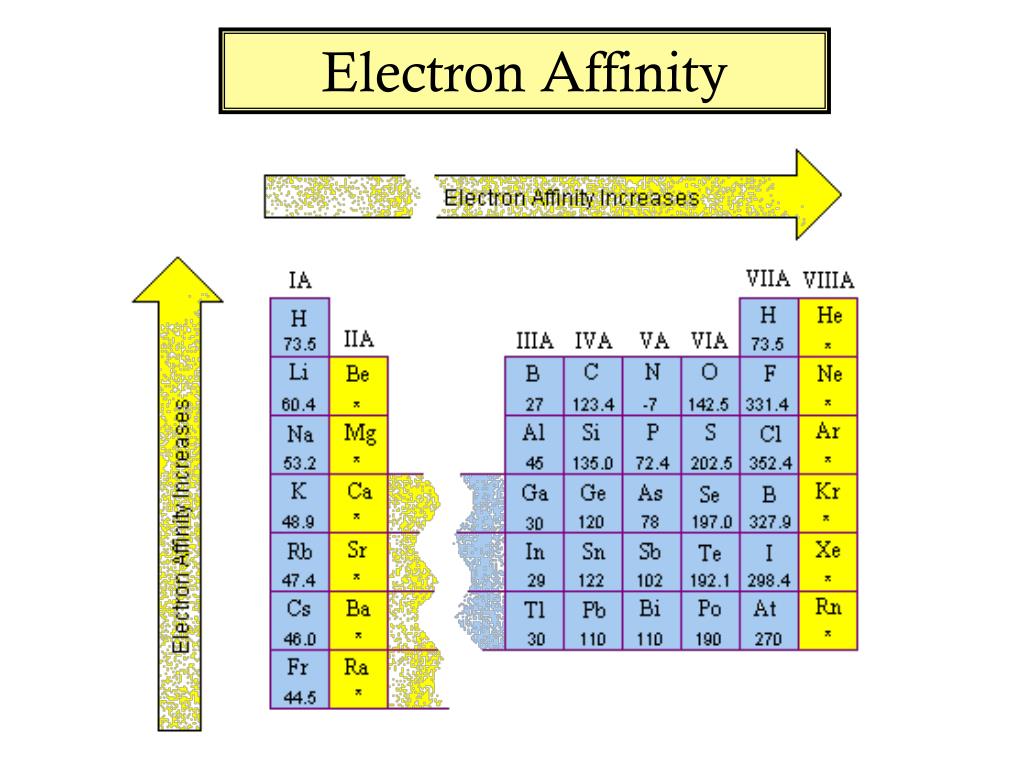
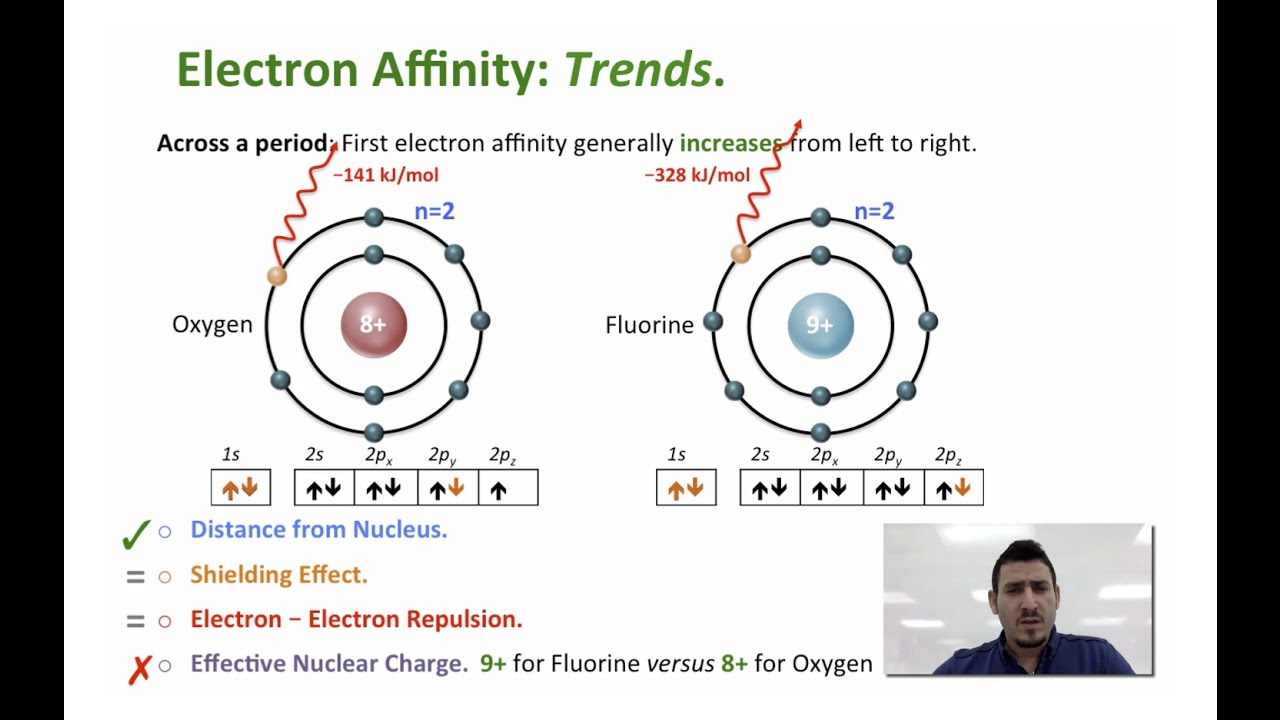
Metals are more likely to lose electrons than they are to gain them. Thus, the top right elements (except for the noble gases) have the highest electron affinities - the greatest energy released when an electron is added. Where is electron affinity highest on the periodic table?Įlectron affinity increases towards the top of the periodic table and towards the right. If an element gains an electron, this creates a negatively charged atom called an anion.ġ. In every element, an equal number of negatively charged electrons are present in shells surrounding the nucleus. Therefore, chlorine has a higher electron affinity than fluorine, and this orbital structure causes it to have the highest electron affinity of all of the elements.Refresher: Elements have positively charged protons in their nucleus that give them a particular nuclear charge. Since chlorine's outermost orbital is a 3p orbital, there is more space, and the electrons in this orbital are inclined to share this space with an extra electron. Because of this, any new electron trying to attach to fluorine experiences lower electron affinity from the electrons already living in the element's 2p orbital. Fluorine is a small atom with a small amount of space available in its 2p orbital. Number of protons and size of atom: Halogens are smaller atom compared to other elements in same horizontal lines in the modern Periodic table.Īs fluorine sits atop chlorine in the periodic table, most people expect it to have the highest electron affinity, but this is not the case. The electron affinity of an atom or molecule is defined as the amount of energy released or spent when an electron is added to a neutral atom or molecule in the gaseous state to form a negative ion.Įlectron affinity of an elements depends on certain factor like

